Statistics and Programming
From conception of study to dissemination of results, statistics and programming play a key role in clinical research.
Our team can provide expertise and knowledge
throughout every step of your studies
Our teams contribute to the set-up and monitoring of clinical trials/studies in collaboration with the study team, ensure the integrity and accuracy of your data, provide advice and expertise on data exploitation, presentation and interpretation of results.
In accordance with ICH (International Conference on Harmonisation of technical requirements for registration of pharmaceuticals for human use) Good Clinical Practices (GCP) or other GXP such as Good Manufacturing Practices (GMP), other international guidelines (e.g. ISO), working group recommendations (e.g. CDISC or Good Programming Practices), and respecting deadlines, quality, regulatory requirements.
Statistics capabilities
- Input in study design , sample size or power calculations Statistical section of any study document (eg, protocol, study report)
- Randomization Statistical review of study documents (eg, Case Report Form, protocol, study risk management plan, IRT specifications, study report…)
- Statistical analysis plans , including shell TL&Fs (Tables, Listings and Figures)
- Data Monitoring Committee (DMC): charter, SAP, shell TLFs
- Conduct and interpretation of statistical analysis
- Meta-analyses
- Statistical report
- Clinical Study report
- Trial disclosure
- Statistical support to Market Access (Review of Payer documents, Preparation of Payer AdBoard, statistical support for internal decision making)
- Regulatory agency discussions Support for publications, abstracts , posters, manuscripts
Programming capabilities
- Review of study/project documents (eg, Case Report Form, Statistical Analysis Plan, Protocol…).
- Development and validation of derived datasets (SDTM, ADaM CDISC format, legacy, pools)
- Development of documentation: specifications (Metadata file, define.xml, cSDRG, ADRG, ARM)
- Validation of datasets, Define.xml by using QC and Pinnacle 21
- Programs in SAS, R, RShiny to meet the study / project needs
- Statistical outputs: tables, listings, figures
- Package for submission (FDA, EMA,..)
- Complementary analyses to support HTA (Health Technology Assessment) submission
Our delivery methods

Technical assistance
Technical support, advice and expertise

Platform
A range of services dedicated to your activities driven by our team

Projects
Performance with commitment to results
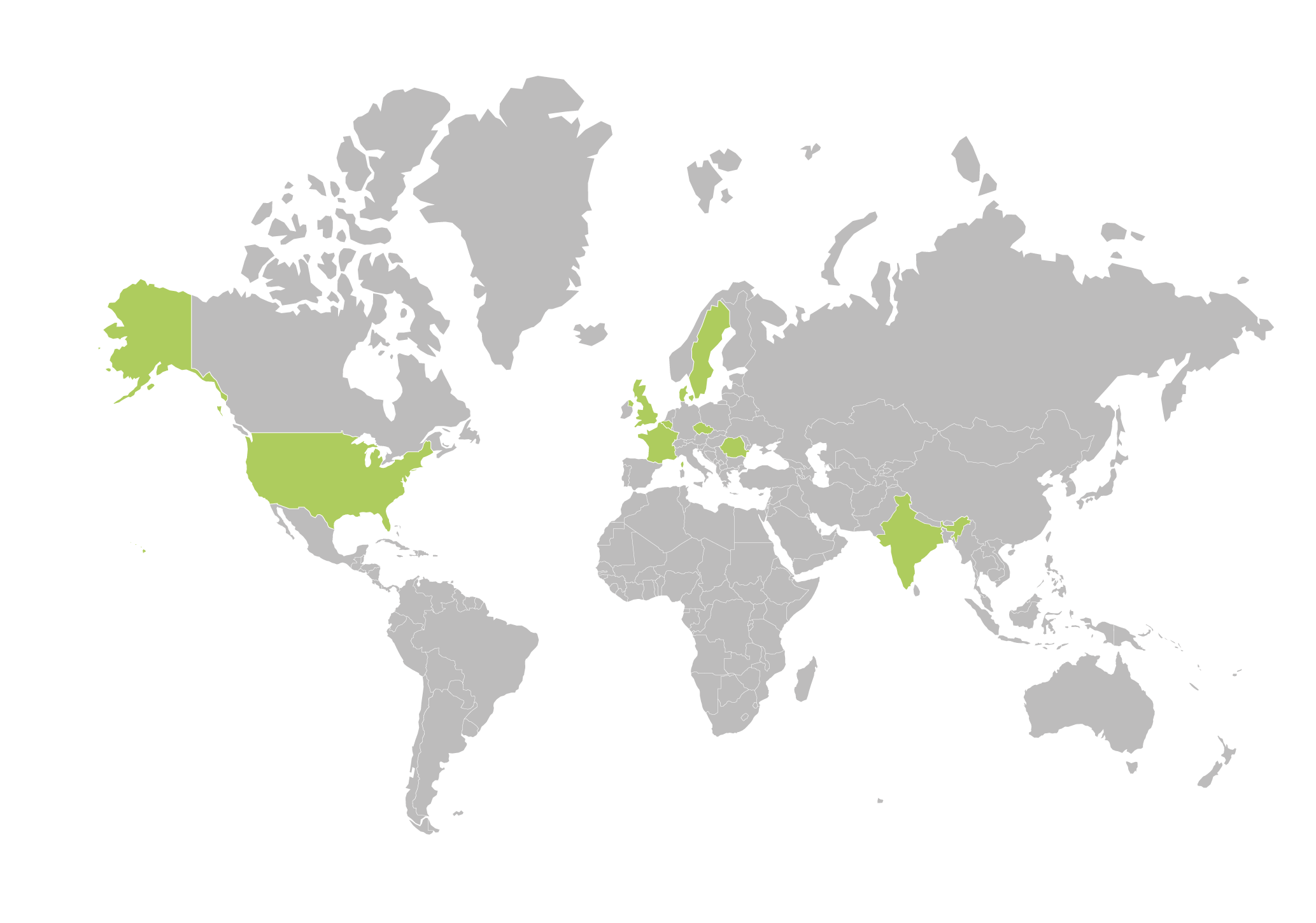
Aixial Group around the world
Aixial Group can help you around the world. We provide services onshore, nearshore, and offshore to ensure that your solution is customized to your specific needs.
Our sectors

Pharmaceutical

Food
Biotech & Vaccines

Medical devices

Veterinary

Cosmetic
How can we support your next project?

OTHER WAYS WE SUPPORT YOU
